
Get SAS A00-281 Exam Dumps
SAS Clinical Trials Programming Using SAS 9 Accelerated Version Exam Dumps
This Bundle Pack includes Following 3 Formats
Test software
Practice Test
Answers (PDF)

A00-281 Desktop Practice
Test Software
Total Questions : 99

A00-281 Questions & Answers
(PDF)
Total Questions : 99

A00-281 Web Based Self Assessment Practice Test
Following are some A00-281 Exam Questions for Review
A patient received at least one dose of study medication prior to withdrawing from a study. Which analysis population would always include this patient?
Identify the data structure with the following characteristics:
* Contains one or more records per subject, per analysis parameter, and per analysis timepoint.
* May be derived from findings, events, interventions and special-purpose SDTM domains, or other ADaM datasets.
* A record can represent an observed, derived, or imputed value required for analysis.
This question will ask you to provide a missing option. The following program is submitted to create a transport file for multiple data sets:
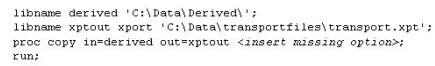
Which option will limit the files that are copied to type data?
The following SAS program is submitted:

Which alternative program creates an equivalent BASE_BP data set?
Which LIBNAME statement is valid?
Unlock All Features of SAS A00-281 Dumps Software
Types you want
pass percentage
(Hours: Minutes)
Practice test with
limited questions
Support