
Get SAS A00-280 Exam Dumps
SAS Certified Clinical Trials Programmer Using SAS 9 Exam Dumps
This Bundle Pack includes Following 3 Formats
Test software
Practice Test
Answers (PDF)

A00-280 Desktop Practice
Test Software
Total Questions : 99

A00-280 Questions & Answers
(PDF)
Total Questions : 99

A00-280 Web Based Self Assessment Practice Test
Following are some A00-280 Exam Questions for Review
A Statistical Analysis Plan (SAP) defines the selection process for baseline records. This instructs the programmer to choose the last non-missing analyte value prior to first study drug administration (date/time).
The DEMO data set contains the date/time of first study drug administration for subject:
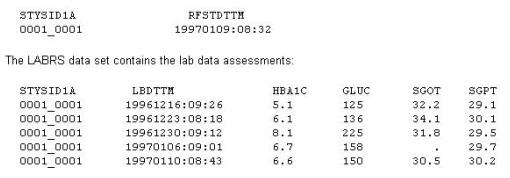
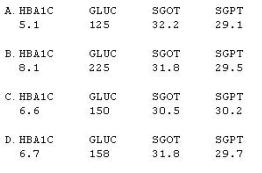
What will be the resulting baseline values, as selected per the SAP instructions?
Which statement correctly describes an aspect of a Phase II clinical trial?
Which CDISC filename contains the following items?
* Variable attributes
* Controlled terminology
* Computational methods
Enter your answer in the space below (Case is ignored. Do not add leading or trailing spaces to your answer.).

Where would you store a value collected on a case report form but not defined in an SDTM domain?
The purpose of the ADaM model is to provide a framework that:
Unlock All Features of SAS A00-280 Dumps Software
Types you want
pass percentage
(Hours: Minutes)
Practice test with
limited questions
Support